 丂丂丂丂
丂丂丂丂丂
Olin丂丂丂http://www.olin.com/about/history.asp丂丂
In October of 1996, Olin
announced a series of strategic initiatives that were designed to
create a stronger, more focused and more valuable company. To
begin with, Olin spun off to shareholders our former Ordnance and
Aerospace divisions as Primex Technologies, Inc.
We also announced the sale of our TDI and ADI isocyanates
businesses to Arco Chemical for
$565 million in cash. While Olin was a leading producer of TDI in
North America, we neither had a presence in important
complementary products such as MDI, nor a strong propylene oxide
position for flexible polyols. We also sold our surfactants
businesses at our chemicals' complex in Brandenburg, Ky. to BASF
and Pilot Chemical Company.
Olin used the proceeds from the TDI sale and other divestments to
repurchase Olin common stock, to pay down debt, and to purchase DuPont's 50% share in
Niachlor -- a joint venture
chlor alkali plant in Niagara Falls, N.Y. The company also
invested in a new
chlor alkali plant in McIntosh, Alabama. The new Sunbelt plant was built for $200
million as a joint venture between Olin and Geon, a major Olin customer in the polyvinyl
chloride (PVC) market. Geon takes 100% of the plant's chlorine
for its use in making PVC resins, while Olin in turn markets the
plant's high-purity caustic soda.
Once these strategic actions were completed, Olin had what
amounted to two different sets of businesses with entirely
different dynamics. On the one hand, the company's specialty
chemicals businesses were driven by the need to supply customers
with tailored chemicals and technology and a high level of
technical customer support. On the other hand, Olin's more
traditional basic materials businesses competed more on price and
sold into more mature, slower growth markets.
In mid-1998, Olin announced that it would spin off its specialty
chemical businesses as a separate, publicly traded company. The purpose of the spin off was to free
each of the new companies -- Olin and the spun off company -- to
focus exclusively on growing its own businesses and competitive
advantages. On February 8, 1999, Olin spun off its specialty
chemical businesses as Arch Chemicals, Inc.
Around that time, every Olin stockholder received one share of
Arch Chemicals stock for every two shares of Olin stock held.
Following the spin off of Arch Chemicals, Olin became a $1.5
billion company that is a leading North American producer of
copper alloys and other metals, ammunition, and chlorine and
caustic soda. One of the world's best basic materials businesses,
the company today is comprised of three divisions -- Olin Brass,
Winchester (both of which are headquartered in East Alton, Ill.)
and the Chlor Alkali Products Division, which is headquartered in
Cleveland, Tenn.
Olin Brass is the world's leading developer of high
performance copper alloys and is the U.S. market share leader in
copper and copper alloy strip. Winchester is the world's largest ammunition
producer and a leading producer of small caliber ammunition in
North America. And Chlor Alkali Products is the largest supplier of chlorine and
caustic soda in the eastern United States and the fourth largest
nationwide. Today, Olin has approximately 6,700 employees,
principally in North America.
We at Olin are excited to be writing a new chapter in our long,
proud history. In the months and years ahead, we will fulfill our
bright promise by capitalizing on our leadership positions in
copper alloys, ammunition and chlorine and caustic soda. We'll do
so by intensely focusing on customer needs, aggressively managing
costs and pursuing synergistic growth opportunities.
 丂丂丂丂
丂丂丂丂
AstraZeneca PLC was formed on 6
April 1999 through the merger of Astra AB of Sweden and Zeneca Group PLC of the UK. The merger represented the
combination of two companies with similar science-based cultures
and a shared vision of the pharmaceutical industry.
The AstraZeneca Board believes that the merger will improve the
combined companies' ability to generate long-term growth and
shareholder value through:
Global power & reach in sales and marketing
丒丂Major primary care
presence, particularly in gastrointestinal, cardiovascular and
respiratory medicine
丒丂Leading position in a
number of specialist/hospital markets, including oncology and
anaesthesia
丒丂Ability to deliver the
potential of existing and future products through the power and
reach of a combined global sales and marketing resource
丒丂Widespread class
coverage in key therapy areas, such as cardiovascular and
respiratory disease, due to complementary nature of products
Stronger R&D platform for innovation led growth
丒丂Substantial research
and development (R&D) expenditure - over $2.5 billion
annually
丒丂Strong combined
development pipeline
丒丂Potential for further
strengthening of the pipeline by enhanced discovery and
development capability through greater scale and focus on
selected areas and technologies - in particular, complementarity
in cardiovascular, respiratory, central nervous system and
anaesthesia/ analgesia R&D should help to deliver significant
benefits
丒丂Research partnerships -
to supplement its own R&D expertise, AstraZeneca will
continue to work with external partners, such as academic
institutions and other pharmaceutical companies, to broaden its
range of early exploratory research and to allow access to a
wider base of scientists and new technologies
Greater financial strategic flexibility
丒丂Financial strength and
scale to give AstraZeneca's management greater strategic
flexibility to drive long-term earnings growth
丒丂Substantial operational
efficiencies resulting in cost savings
丂丂丂http://www.astrazeneca.com.au/AboutUs/1990s.htm
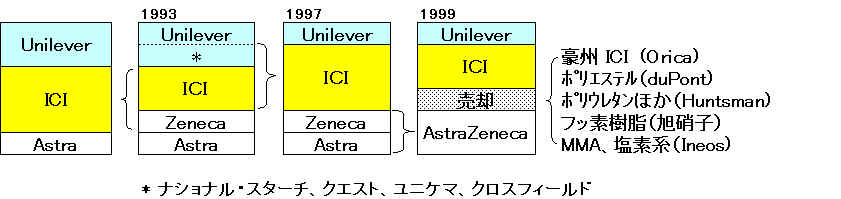
At Zeneca
The 1990s was a time of great change for ICI Pharmaceuticals with
the birth of Zeneca and then AstraZeneca, one of the world's
leading pharmaceutical companies, at the end of the decade.
By the 1990s, ICI Pharmaceuticals employed more than 12,000
people. It had 21 production sites and 150 sales offices around
the world, and major research laboratories in the USA, France and
the UK.
In 1993, ICI
demerged three of its businesses (Pharmaceuticals, Agrochemicals
and Specialties) to form a separate company, Zeneca.
Zeneca continued to build on its pharmaceutical inheritance in
cardiovascular, anti-cancer, anti-infective and anaesthetic
medicines. Research and effective licensing strategies in
respiratory and central nervous system medicine also brought
great rewards.
In 1995, Casodex (bicalutamide) was launched for the treatment of
advanced prostate cancer. A new long-acting 3-month depot
formulation of Zoladex (goserelin acetate) was also released,
which represented a significant development in drug delivery for
which Zeneca received the UK Queen's Award for Industry.
In respiratory medicine, the leukotriene receptor antagonist
Accolate (zafirlukast) was launched in 1996 for the treatment of
asthma. Leukotriene receptor antagonism represents the first
novel approach to asthma therapy in 20 years.
In central nervous system medicine, 1997 saw the release of
Seroquel (quetiapine), an atypical anti-psychotic agent, for the
treatment of schizophrenia and other psychotic disorders. Zomig
(zolmitriptan), a selective 5-hydroxytryptamine1B/1D
(5-HT1B/1D) receptor agonist, was also launched for the
treatment of acute migraine (licensed from Glaxo Wellcome).
伀
On 6 April 1999, Astra officially merged with Zeneca to form AstraZeneca. The merger represented the combination of two companies with science-based cultures and a shared vision of the pharmaceutical industry.
仾
At Astra丂of Sweden丂丂
Early in this decade, the
organisation of Astra's international marketing was expanded, and
several licensing agreements renegotiated to give the company
control over the marketing of its products in major markets.
Local marketing companies were acquired in Italy and Spain in
1990 and 1991. In 1994, Astra invested approximately SEK 8
billion in raised ownership (90%) in Astra Japan and in the
formation of Astra Merck Inc. in the USA.
The largest expansion of Astra's research and development in
modern times was carried out in 1995 with acquisition of new
research facilities in the UK (Astra Charnwood) and USA (Astra
Arcus USA).
In 1996, Losec (omeprazole) achieved the position as the world's
biggest-selling pharmaceutical. The success of the Turbuhaler
inhalation system for Pulmicort (budesonide), Oxis (formoterol)
and Rhinocort (budesonide) respiratory agents continued.
Astra shares were listed on the New York Stock exchange from May
1996.
In 1997, Astra launched Atacand (candesartan cilexetil), a new
angiotensin II type 1 (AT1)-receptor blocker, for the treatment
of hypertension to further support the broad portfolio of
cardiovascular drugs. In July 1998, the Astra Merck relationship
in the USA was restructured to form Astra Pharmaceuticals, L.P.,
and in the same year the advanced formulation of Losec, Losec
MUPS (Multiple Unit Pellet System) tablets, reached its first
markets.
1997/7/8丂丂丂丂http://www.ici.com/pressoffice/news/08_07_1997.htm
ICI Completes Acquisition of Unilever Speciality
Chemicals Businesses
ICI has completed the acquisition of the Speciality Chemicals
businesses of Unilever for a debt free price of $8 billion (亽4.9 bn). The businesses are:-
丂丒丂National Starch, a
world leader in industrial adhesives and number one in specialty
starches
丂丒丂Quest, one of the
world's leading fragrance, food ingredient and flavours companies
丂丒丂Unichema, a global
leader in oleochemicals
丂丒丂Crosfield, a major
producer of silicates, zeolites and silicas
ICI Chairman Sir Ronald Hampel welcomed the businesses into the
ICI Group. "The acquisition is a major milestone in
delivering a new ICI for the new century, "he said. "It
will move ICI's product portfolio more towards market driven
businesses supported by outstanding selling skills and science.
"The portfolio of businesses in ICI is well positioned to
develop and deliver sustainable profitable growth over the coming
years."
The deal was first announced in May 1997.
丂Pharmacia Corporation has emerged as a leading global pharmaceutical enterprise with a robust product portfolio, wide geographic reach and balance, and a $2 billion research engine to drive new product flow.
丂The roots of Pharmacia
Corporation date back almost 150 years to 1853 when a leading
Italian pharmacist, Carlo Erba, started his own company, which
later became Farmitalia Carlo Erba. This company would later
unite with Kabi Pharmacia, which began in 1931. These two
companies, along with Pharmacia Aktiebolag, form the three main
points of origin for Pharmacia AB, a Swedish-based company.
丂In the United States,
Pharmacia traces its roots back to 1886. It was then that W.E.
Upjohn, M.D., established The Upjohn
Pill and Granule Company of Kalamazoo, Michigan, (USA). The
company continued its growth throughout the 19th century,
eventually evolving into an innovative, international company.
In 1995, Pharmacia
& Upjohn was formed through the merger of Pharmacia AB and
The Upjohn Company. Pharmacia
& Upjohn became a global provider of human healthcare
products, animal health products, diagnostics and specialty
products. In 1998, Pharmacia & Upjohn relocated its global
headquarters from the United Kingdom to New Jersey (USA). In
September 1999, the company established its global headquarters
on a 70-acre campus in Peapack, New Jersey. This site is now the
management and pharmaceutical headquarters for Pharmacia
Corporation.
丂Monsanto was formed in 1901 when a high-school
dropout and entrepreneur, John F. Queeny, founded the company in
St. Louis, Missouri (USA), and began producing saccharin, an
artificial sweetener. Together with Dr. Louis Veillon, a chemist
from Switzerland, Queeny helped grow Monsanto into one of the
largest chemical companies in the United States. In 1927,
Monsanto registered as a public company and stock was first
issued on Oct. 10, 1929. Following saccharin, other products such
as aspirin, caffeine, and vanilla were being produced.
丂As Queeny and his son,
Edgar, grew Monsanto by aggressive licensing and acquisitions, as
well as by in-house research, another company also was
prospering. G.D.
Searle & Co., formed in
1888 by a young druggist named Gideon Daniel Searle, was an
innovative company developing many "firsts." These
include the first bulk laxative, the first motion-sickness drug,
the first oral contraceptive, the first once-a-day calcium
channel blocker for the treatment of hypertension, and many other
innovative products. Likewise, Searle discovered and introduced
aspartame, a hugely successful sugar substitute. G.D. Searle
& Co. had its headquarters in Skokie, Illinois. In 1985, G.D. Searle & Co.
became the pharmaceutical unit of Monsanto.
(儌儞僒儞僩幮偲傾儊儕僇儞丒儂乕儉丒僾儘僟僋僣幮偺崌暪 仺丂攋択)
丂Monsanto further defined
itself when, in August 1997, shareowners of the company approved
the spin-off
of its chemical business, Solutia Inc. In less than 100 years, Monsanto
established itself as a proven leader in the agricultural and
pharmaceutical businesses.
丂On April 3, 2000, Monsanto and Pharmacia
& Upjohn completed a merger creating a dynamic and powerful new
competitor in the pharmaceutical industry.丂This new company乕Pharmacia Corporation乕is a high-performance competitor. Working
in teams as part of a global organization, we are building one of
the best-managed companies in the industry. We are responding as
a partner to meet the diverse needs of the people we serve,
through a flow of innovative medicines, products and services. By
doing so, we are delivering best-managed performance for our
shareowners: superior long-term growth and profitability.
spin-off of its remaining 84% ownership of Monsanto to its current shareholders. 丂仺spin-off
Pfizer to acquire Pharmacia Corporation
As Pharmacia Completes Monsanto
SpinOff, Monsanto CEO Says Company Retains Focus and Strategic
Direction
As previously announced, at the close of business today Pharmacia
Corporation will distribute its 84-percent stake in Monsanto
Company to Pharmacia shareowners via a special stock dividend.
This distribution will complete Pharmacia's spinoff of Monsanto
and establish Monsanto as a 100-percent publicly traded company.
In marking the occasion, Monsanto President and Chief Executive
Officer Hendrik A. Verfaillie said Monsanto's focus would remain
on its long-term commitment to agriculture and the strategy it
has built around its customer base. "Today marks an
important milestone, affirming Monsanto's place as a company
solely dedicated to agriculture," said Verfaillie.
"However, this is just one milestone in a larger evolution.
We've assembled a company around our integrated solutions
strategy, and we believe that our strategy uniquely positions us
as a leader in the agricultural industry." Monsanto's
customer-driven integrated solutions strategy includes seeds,
biotechnology traits and agricultural chemistry. The company
enhances its strengths in these three individual elements by
combining them to provide integrated solutions for farm
productivity and food quality, Verfaillie said.
"In the past few years, commodity agriculture has been
besieged by challenges ranging from depressed prices to
extraordinary weather," said Verfaillie. "Our intention
is to focus our efforts on solutions that will improve farmers'
bottom-line productivity. This is something we see as a timeless,
underlying benefit that would be recognized in any market
condition." In addition to Monsanto's business commitment to
farmers, Verfaillie also has committed the company to the New
Monsanto Pledge, a code of conduct for doing business. The Pledge
includes five areas of commitment: delivering benefits to farmers
and the environment; dialogue with interested parties;
transparency with information and regulation; respect for other
points of view; and sharing of technology.
In connection with the spinoff of Monsanto from Pharmacia,
Verfaillie also announced that Christopher J. Coughlin, of
Morristown, New Jersey, will resign from the Monsanto board of
directors effective today. Coughlin, 50, is executive vice
president and chief financial officer of Pharmacia and has been a
member of the Monsanto board since March 2000. "We thank
Chris for his service and dedication to Monsanto shareowners
during the past two years on our board," said Verfaillie.
This change to the Monsanto board brings the number of directors
to nine. Verfaillie said Monsanto will look to add an independent
director to the Monsanto board in the future. Monsanto Company
(NYSE: MON) is a leading global provider of technology-based
solutions and agricultural products that improve farm
productivity and food quality. For more information on Monsanto,
see: www.monsanto.com.
2003/4/17丂Aventis
Aventis Reduces its Shareholding in
Rhodia, Retains Economic Interest
Strasbourg, France -Aventis
announces today the reduction of its stake in Rhodia to
15.3% from 25.2% following a
definitive sale and purchase agreement with Credit Lyonnais
concerning 17.8 million Rhodia shares (40% of its Rhodia
shareholding). As a result of this transaction, Aventis will hold
27.5 million Rhodia shares and Credit Lyonnais will have acquired
9.9% of Rhodia乫s share
capital. The sale will be formally completed by the end of April.
The price will be the market price of the Rhodia shares on the
day prior to the closing of this transaction.
Separately, Aventis and Credit
Lyonnais have reached agreement whereby Aventis will retain an
interest in the future evolution, whether positive or negative,
of the Rhodia share price, for a period of up to five years.
Commenting on this transaction,
Patrick Langlois, Vice Chairman and Chief Financial Officer of
Aventis, said: 乬This
transaction is an important step in our continued focus on our
core pharmaceutical activities
and reaffirms our commitment to our shareholders to exit the non-core
industrial activities. In
addition, this transaction is a first step to comply with the
requirement to
reduce the Aventis shareholding in Rhodia to below 5% by April
2004, pursuant to the 1999 agreement with EU and US antitrust
authorities. The structure of
this transaction enables us to retain the upside potential as
valuations of chemical companies improve in conjunction with a
recovery in the economic cycle and as Rhodia executes its乫
business plan. Of course, we will
continue to seek ways to divest significant portions of our
remaining shareholding in Rhodia,乭 concluded Langlois.
In addition to Rhodia, Aventis holds
a 12% stake in the Swiss specialty chemicals company Clariant.
Given the recent decline in the market value of these companies,
Aventis intends to book a provision in its first quarter results
to reflect the current market value of these companies relative
to their book value.
About Aventis
Aventis is dedicated to treating and
preventing disease by discovering and developing innovative
prescription drugs and human vaccines. In 2002, Aventis generated
sales of ? 17.6 billion, invested ? 3.1 billion in research and
development and employed approximately 71,000 people in its core
business. Aventis corporate headquarters are in Strasbourg,
France. For more information, please visit: www.aventis.com
Statements in this news release
other than historical information are forward-looking statement
subject to risks and uncertainties. Actual results could differ
materially depending on factors such as the availability of
resources, the timing and effects of regulatory actions, the
strength of competition, the outcome of litigation and the
effectiveness of patent protection. Additional information
regarding risks and uncertainties is set forth in the current
Annual Report on Form 20-F of Aventis on file with the Securities
and Exchange Commission.
Notes to editors:
On the occasion of the creation of
Aventis in 1999, Aventis agreed to progressively dispose of its
stake in Rhodia pursuant to the antitrust review process carried
out by the European Commission in Europe and by the Federal Trade
Commission in the United States.
Specifically, in October 1999,
Aventis decided to sell immediately part of its stake in Rhodia
via an equity offering (thus reducing its interest in Rhodia from
67.3% to 25.2%) and to simultaneously issue Exchangeable Bonds
with respect to its remaining interest in Rhodia.
On November 28, 2002, Aventis
announced a cash tender offer to buy back the Exchangeable Bonds
in order to enhance its flexibility for the disposal of its stake
in Rhodia.
On January 17, 2003, all the
Exchangeable Bonds had been repurchased and cancelled and the
shares, which were underlying the tendered Bonds, are kept by the
Escrow Agent until Aventis completes their disposal. As a
consequence, the shares sold today to Credit Lyonnais will be
released by the Escrow Agent immediately prior to their transfer
to Credit Lyonnais.
May 14, 2003 Financial Times
In pole position/ PKN Orlen, Poland.
PKN Orlen is the largest chemical establishment in Poland. Its
history is indicated. Its turnover in 2002 was Zloty 16.8 bn (EUR
3.9 bn/$4.3 bn), which was 1.2% lower than in 2001. Earnings
before interest rose by 19% to Zloty 735 M.
The company has the largest refinery in Poland and it is the most efficient of its type
in Europe It makes feedstock for the integrated petrochemicals
and polymers plants.
Uhde is upgrading the aromatics plant by the addition of a new
unit using the Uhde Morphylane technology, coming on stream in 2Q
2004 to make 400,000 tonnes/y of benzene and toluene.
PKN Orlen holds 75% of Zaklady Azotowe Anwil, which has the biggest polyvinyl chloride (PVC) plant in Poland, and also makes nitrogen
fertilizers and polyethylene (PE) packaging, using ethylene from
PKN Orlen.
PKN Orlen made 2 m tonnes of petrochemicals in 2002 which was 30%
more than the 1.6 m tonnes it made in 2001. Demand in Poland for
polyolefins is increasing at 8-13%/y and half of the supply comes
from imports.
PKN Orlen and Basell have established a 50:50 joint venture (jv) Basell Orlen Poliolefins to construct a 400,000 polypropylene (PP) plant and a 320,000 tonnes/y high
density polyethylene (HDPE)
plant using the Spheripol and Hostalen Basell technology, and
starting up in 1H 2005.
PKN is expanding its ethylene capacity from 360,000 tonnes/y to 660,000
tonnes/y and its propylene capacity from 240,000 tonnes/y to
445,000 tonnes/y to supply the jv.
The PKN low density polyethylene (LDPE)
plant with a capacity of 150,000 tonnes/y will be taken into the
jv and the capacity will be reduced to 105,000 tonnes/y when the
HDPE plant is started up. The PKN PP plant has been recently
expanded to 140,000 tonnes and it will be part of the jv,
although the old PP plant will be closed. The advantages of the
jv are discussed.
http://www.millenniumchem.com/About+Us/Profile/Profile_EN.htm
Millennium
Chemicals (MCH - NYSE) is one of the leading chemical companies
in the world.
We supply millions of pounds of chemicals each year to
manufacturers for use in thousands of consumer and industrial
products. In short, we work hard to improve the fundamentals of
daily life.
Millennium Chemicals is乧
| 丒 | The world's second-largest producer of titanium dioxide (TiO2) and the largest merchant seller of titanium tetrachloride (TiCl4) in North America and Europe. |
| 丒 | A leading producer of fragrance chemicals |
| 丒 | The second-largest producer of acetic acid and vinyl acetate monomer in North America, and through its partnership interest in LaPorte Methanol Company, LP, a partner in a leading U.S.A. producer of methanol. |
| 丒 | A 29.5% owner of Equistar Chemicals, LP, the second-largest producer of ethylene and the third-largest producer of polyethylene in North America. |
Millennium Chemicals' corporate headquarters are located in Red Bank, New Jersey. Millennium Chemicals' global network includes 4,000 employees on five continents. View our directory for a complete listing of office and manufacturing locations.
丂
Ciba to focus on Asia for all new plants
Asia's fundamental importance to
the growth plans of Swiss specialty chemicals company Ciba was
underlined here on Tuesday by chairman and chief executive Armin
Meyer when he said all future capacity expansions would be
concentrated in the region.
Meyer, who was speaking at the company's annual results press
conference, said investments in the traditional regions of Europe
and the US would focus primarily on maintaining world-class
standards and process improvements.
"We don't plan any new plants in Europe or the US,"
said Meyer. "It doesn't make sense."
Meyer stressed that Ciba has very modern, well-equipped plants in
Europe and North America. "Of course we will modernise and
eliminate bottlenecks," he said. But all major new
greenfield plants will be located in Asia, with China the most
likely location.
Ciba has budgeted around SF250m ($198m/Euro160m) on annual
capital expenditure over the next two to three years, said Meyer.
About 60% will be spent on maintaining or improving environmental
health and safety, debottlenecking and further automation.
The remaining 40% will be invested in new production lines,
mainly in the home and personal care, plastic additives and
coating effects segments.
In defending the size of Ciba's capital expenditure budget, Meyer
said it did not include computers, software or research and
development (R&D). "And you need less money to build in
Asia," he added.
Meyer also explained that by using standardised technology and
systems, the cost of new plants could be reduced.
2004/2/17 Novartis 丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂丂擔杮
Novartis completes strategic
acquisition to strengthen number two position in global medical
nutrition business
http://dominoext.novartis.com/NC/NCPRRE01.nsf/44aff02a639be034c1256b4b007b5f4d/f56ae6505bad1277c1256e3d00226082?OpenDocument
丒 New presence in US retail
medical nutrition channel, access to Japanese market and strong
brand portfolio benefit Novartis Medical Nutrition
Novartis has completed the acquisition of Mead Johnson & Company's global adult medical
nutrition business in a USD
385 million cash transaction. No divestments were required to
obtain regulatory approval of the deal, which officially closed
February 13, 2004.
The acquisition adds strong brands like Boost® , Isocal® and Ultracal® to Novartis Medical Nutrition's portfolio
and expands its ability to meet the medical nutritional needs of
a growing outpatient and ageing population.
Novartis Medical Nutrition now enjoys a strong presence in the
fast-growing US retail channel for medical nutrition products, a
platform in the Japanese market and increased opportunities to
build its existing institutional medical nutrition business.
Announced on December 16, 2003, the acquisition includes the
brands, trademarks, patents and intellectual property assets of
Mead Johnson & Company's adult medical nutrition business.
Novartis Medical Nutrition, with global sales of USD 815 million
in 2003 (including Nutrition & Sante), offers a complete
range of enteral (tube feeding) and oral nutrition products and
devices tailored to the varying needs of patients and healthcare
professionals. The product range encompasses supplements, which
are taken orally, as well as other products administered through
tube feeds and specific medical devices. Its key brands include
Isosource® , Novasource®
, ResourceR, Impact® and Compat® .
About Novartis
Novartis AG (NYSE: NVS) is a world leader in pharmaceuticals and
consumer health. In 2003, the Group's businesses achieved sales
of USD 24.9 billion and a net income of USD 5.0 billion. The
Group invested approximately USD 3.8 billion in R&D.
Headquartered in Basel, Switzerland, Novartis Group companies
employ about 78 500 people and operate in over 140 countries
around the world. For further information please consult http://www.novartis.com.
Mead Johnson Nutritionals
http://www.meadjohnson.ca/about/Mead_Johnson_Nutritional_Heritage.htmMead Johnson Nutritionals' worldwide leadership in nutrition can be traced back nearly a century - to Edward Mead Johnson himself, the company's namesake and founder. In 1888, the life of E. Mead's baby son, Ted, was in danger because he was not thriving on his feedings and had to be fed a cooked "gruel" mixture. Years later, memory of this experience would inspire E. Mead to develop a product that would eventually lead to today's worldwide leader in infant formula sales, of Enfamil and Enfalac.
E. Mead first co-founded Johnson & Johnson with his brother. After a third brother joined the company, E. Mead decided to pursue other interests and formed the American Ferment Company, which he renamed Mead Johnson & Company in 1905. As he started his company, he explored a variety of products; however, his strongest interest was in scientifically based nutritional products.Mead Johnson & Company became a wholly owned subsidiary of Bristol-Myers Company (now Bristol-Myers Squibb Company) in 1967. Bristol-Myers Squibb is a diversified worldwide health company. It is a leading maker of innovative therapies for cardiovascular, metabolic and infectious diseases, central nervous system and dermatological disorders, and cancer, as well as osmotic care, wound management, nutritional supplements, and infant formulas.
擔杮偵偍偗傞乽惉恖岦偗堛椕梡塰梴怘昳乿帠嬈偺堏娗偵偮偄偰
http://www.novartis.co.jp/pdf/pr040218-2.pdf
丂僲僶儖僥傿僗僼傽乕儅姅幃夛幮乮杮幮丗搶嫗搒峘嬫丄戙昞庢掲栶幮挿丗捠嬝夒峅乯偼丄僲僶儖僥傿僗幮乮杮幮丗僗僀僗僶乕僛儖乯偑丄儈乕僪僕儑儞僜儞幮乮杮幮丗暷崙僀儞僨傿傾僫廈丅暷崙僽儕僗僩儖丒儅僀儎乕僘僗僋僀僽幮偺巕夛幮乯偺乽惉恖岦偗堛椕梡塰梴怘昳乿帠嬈傪悽奅揑偵庢摼偟偨偲偺敪昞偵婎偯偒丄擔杮偵偍偗傞摨帠嬈偺堏娗偵偮偄偰丄師偺偲偍傝寛掕偟傑偟偨偺偱偍抦傜偣偄偨偟傑偡丅
丂2004擭2寧14擔傛傝丄儈乕僪僕儑儞僜儞姅幃夛幮乮僽儕僗僩儖丒儅僀儎乕僘僗僋僀僽乮桳乯偺100亾巕夛幮乯偺忋婰帠嬈偑丄偁傞堦掕婜娫丄僲僶儖僥傿僗僼傽乕儅乮姅乯偵堏娗偝傟丄彨棃揑偵丄僲僶儖僥傿僗僌儖乕僾偺拞偺暿夛幮偵堏娗偝傟傞梊掕偱偡丅
丂偙偺帠嬈傪傛傝廳梫偲埵抲偯偗傞僲僶儖僥傿僗偺慻怐偵慻傒崬傑傟傞偙偲偵傛傝丄擔杮偵偍偄偰傕丄崱屻偺帠嬈揥奐偺怴偨側揥朷偑奐偗傞傕偺偲婜懸偝傟偰偄傑偡丅
丂堏娗偝傟傞乽惉恖岦偗堛椕梡塰梴怘昳乿帠嬈偼丄擔杮偵偍偄偰1987擭傛傝昦堾岦偗宱挵塰梴怘昳巗応偵杮奿嶲擖偟丄尰嵼丄宱挵塰梴怘昳巗応偱栺17亾偺僔僃傾傪妉摼偟丄嬈奅戞2埵偺抧埵偵偁傝傑偡丅媫懍側崅楊壔幮夛偵岦偗偰昦堾丄巤愝傪拞怱偵崅偄拲栚傪梺傃偰偄傑偡丅
僲僶儖僥僀僗儊僨傿僇儖僯儏乕僩儕僔儑儞乮堛椕梡塰梴怘昳乯帠嬈晹偵偮偄偰
丂僲僶儖僥傿僗僐儞僔儏乕儅乕僿儖僗晹栧偵偍偗傞僲僶儖僥僀僗儊僨傿僇儖僯儏乕僩儕僔儑儞帠嬈晹偺2003擭搙偺攧傝忋偘偼丄8壄1愮500枩僪儖乮937壄墌乯偱丄姵幰偝傫側傜傃偵僿儖僗働傾偺愱栧壠偺偝傑偞傑側僯乕僘偵墳偊傞丄宱挵乮宱娗乯偍傛傃宱岥偺峀斖埻偵傢偨傞堛椕梡塰梴怘昳偲娭楢堛椕婍嬶傪斕攧偟偰偄傑偡丅
丂惢昳偺斖埻偼宱岥偱愛庢偡傞僒僾儖儊儞僩丄偍傛傃娗偍傛傃摿庩側堛椕婍嬶偵傛傝愛庢偡傞偦偺懠偺堛椕梡塰梴怘昳側偳丄懡婒偵傢偨傝傑偡丅
僲僶儖僥傿僗偵偮偄偰
丂僲僶儖僥傿僗偼丄堛栻昳晹栧偲僐儞僔儏乕儅乕僿儖僗晹栧偐傜側傝丄偦傟偧傟偺暘栰偵偍偗傞悽奅揑儕乕僟乕偱偡丅僲僶儖僥傿僗僌儖乕僾慡懱偺2003擭搙偺攧傝忋偘偼249壄僪儖乮栺2挍8,600壄墌乯偱丄弮棙塿偼50壄僪儖乮栺5,770壄墌乯丄尋媶奐敪傊偺搳帒偼栺38壄僪儖乮栺4,370壄墌乯偱偟偨丅僗僀僗丒僶乕僛儖巗偵杮嫆傪抲偔僲僶儖僥傿僗偼丄栺78,500恖偺幮堳傪梚偟偰偍傝丄悽奅140僇崙埲忋偱惢昳偑斕攧偝傟偰偄傑偡丅徻嵶偼僀儞僞乕僱僢僩傪偛棗偔偩偝偄仺http://www.novartis.com
擔杮宱嵪怴暦丂2004/2/21
奀奜夛幮尋媶丂儘僔儏
崌暪傛傝棙塿棪廳帇丂拞奜惢栻捠偠擔杮帠嬈奼戝
丂婯柾偱偼側偔幙傪捛偆乕乕丅悽奅揑側惢栻嬈奅嵞曇偺偆偹傝傪墶栚偵丄僗僀僗偺惢栻戝庤儘僔儏偑帺幮偺拞妀帠嬈嫮壔傪恑傔偰偄傞丅儔僀僶儖偺僗僀僗惢栻戝庤僲僶儖僥傿僗偐傜偺崌暪採埬傪偐偨偔側偵嫅斲丅堛栻昳偲恌抐栻偺俀晹栧偱懌応傪屌傔傞愴棯偩丅
丂乽偦偆偟偨乮崌廬楢峵偺乯棳傟偵偼嶲壛偟側偄乿丅俀寧係擔丄僗僀僗丒僶乕僛儖偺杮幮偱奐偄偨擭師婰幰夛尒丅僼乕儅乕夛挿寭嵟崅宱塩愑擟幰乮俠俤俷乯偼抐尵偟偨丅
仴崌廬楢峵偲偼堦慄
丂偦偺侾廡娫慜丄暓僜僲僼傿丒僒儞僥儔儃偑傾儀儞僥傿僗偵揋懳揑俿俷俛乮姅幃岞奐攦偄晅偗乯傪敪昞丅懳峈偟偰僲僶儖僥傿僗傕傾儀儞僥傿僗攦廂偵忔傝弌偡偲偺娤應偑媫晜忋偡傞側偳丄悽奅偺惢栻戝庤偺嵞曇婡塣偑堦婥偵崅傑偭偰偄偨丅偩偑儘僔儏偼崌廬払峵偲偼堦慄傪夋偟丄帺庡撈棫傪柧妋偵偟偨丅
丂攚宨偵偼偄偨偢傜偵攧傝忋偘傪奼戝偡傞傛傝丄傑偩傑偩掅偄棙塿悈弨偺岦忋傪桪愭偝偣偨偄偲偺巚偄偑偵偠傓丅俀侽侽俁擭侾俀寧婜偺嵟廔懝塿偼俁侽壄俇俋侽侽枩僗僀僗僼儔儞乮栺俀俇侽侽壄墌乯偺崟帤丅曐桳姅偺娷傒懝傪堦憒偟偰係侽壄僗僀僗僼儔儞偺愒帤偲側偭偨慜偺擭偐傜媫夵慞偟偨丅偩偑僺乕僋偩偭偨俀侽侽侽擭侾俀寧婜偺俉俇壄僗僀僗僼儔儞偲斾傋傞偲丄傑偩俁暘偺侾偩丅
丂僲僶儖僥傿僗偲斾傋偰傕嵎偼柧傜偐丅慜婜偺攧忋崅偼擔杮墌偱栺俀挍俇俆侽侽壄墌偲傎傏偒偭峈偡傞偑丄僲僶儖僥傿僗偺嵟廔棙塿乮栺俆俁侽侽壄墌乯偼夁嫀嵟崅傪峏怴丄儘僔儏偺俀攞偩丅
仴拞妀晹栧傪嫮壔
丂棙塿棪夵慞夵慞僿僐僗僩埑弅傪媫偄偱偄傞丅怴栻奐敪偵敽偆尋媶奐敪旓偼係俉壄僗僀僗僼儔儞偲侾俀亾憹壛丅怴栻敪攧偵敽偆塩嬈旓傕憹偊偨偑丄惢憿尨壙傗堦斒娗棟旓側偳傪戝暆偵埑弅丄媧廂偟偨丅堛栻昳晹栧偺攧忋崅塩嬈棙塿棪偼俀侾丏俋亾偐傜俀俁亾偵岦忋偟偨丅俀侽侽係擭侾俀寧婜偼偙傟傪俀俇亾偵堷偒忋偘傞寁夋偩丅
丂傕偆傂偲偮偺廂塿憹嫮嶔偑拞妀晹栧傊偺乽廤拞偲奼戝乿丅晄嵦嶼偩偭偨價僞儈儞丒僼傽僀儞働儈僇儖晹栧傪嶐擭俋寧枛偱攧媝丅堦曽偱恌抐栻帠嬈偺嫮壔偵椡傪擖傟偨丅
仴巗応偼岲堄揑
丂戝庤摨巑偺崌廬楢峵偼斲掕偡傞偑丄晹栧嫮壔偺偨傔偺彫婯柾側帠嬈攦廂偵偼傓偟傠愊嬌揑丅嶐擭丄僗僀僗偺堛椕婡婍儊乕僇乕偱摐擜昦帯椕梡僀儞僗儕儞億儞僾悽奅俀埵偺僨傿僙儞僩儘僯僢僋傪侾俀壄僪儖偱嶱壓偵廂傔偨丅傑偨丄偙偺傎偳侾係壄僪儖傪搳偠偰丄暷専嵏婡婍儀儞僠儍乕偺傾僀僕僃儞丒僀儞僞乕僫僔儑僫儖偺攦廂傪姰椆偟偨丅
丂俀侽侽俀擭廐偵嶱壓偵廂傔偨拞奜惢栻偲偺楢実岠壥偵傕婜懸傪婑偣傞丅儕儏乕儅僠惈娭愡墛帯椕嵻側偳偺暘栰偱嫤椡娭學傪嫮傔偰偄偔曽恓偱丄棙塿柺偱偺峷專傪婜懸偡傞丅悽奅俀埵偺巗応偱偁傞擔杮偱偺帠嬈奼戝傪慱偄丄拞奜栻傪捠偠偨帠嬈攦廂側偳偵傕慜岦偒偺巔惃傪傒偣傞丅
丂戝庤偲偺崌暪側偳傪斲掕偟棙塿廳帇傪慛柧偵偡傞儘僔儏偺巔惃偵丄巗応偼偍偍傓偹岲堄揑偩丅嵟崅嵿柋愑擟幰偺僼儞僣傿僇乕幏峴栶堳傕乽悽奅偵偼婯柾偱偼側偔幙偑戝帠偩偲峫偊傞搳帒壠傕懡偔偄傞乿偲搳帒壠偺棟夝偑摼傜傟偰偄傞偲嫮挷偡傞丅幚嵺丄懠偺戝庤偲偺崌暪偵慜岦偒側僲僶儖僥傿僗偺姅壙偵斾傋丄嶐擭敿偽埲崀偺栠傝偼儘僔儏姅偵孯攝偑忋偑傞丅
丂僲僶儖僥傿僗偲偺崌暪榖偑扞忋偘偵側偭偰偐傜傕姅壙偼塭嬁傪庴偗偰偄側偄丅僲僶儖僥傿僗偼儘僔儏偺媍寛尃姅偺俁俁丏俁亾傪帩偮偑丄摨幮偺僶僙儔夛挿偼乽偄傑偺偲偙傠攦偄憹偡梊掕偼側偄乿偲偄偆丅儘僔儏懁偵攝椂偡傞宍偱丄崌暪偼椉幮宱塩恮偺崌堄偑慜採偲偄偆巔惃傪偲偭偰偄傞丅
丂偩偑丄儘僔儏偑姅幃偺俁暘偺侾傪儔僀僶儖偵埇傜傟偰偄傞偲偄偆帠幚偼梙傞偑側偄丅戝庤摨巑偺嵞曇偺棳傟偲柍墢偱偄傜傟傞偐偳偆偐偼梊抐傪嫋偝側偄丅